The Engine Powering Automated Cell Processing
BendBio provides the core inertial microfluidics engine for next-generation cell therapy manufacturing systems. Overcome the limitations of legacy platforms with our proven, integration-ready modules for sorting, washing, concentration, and filtration.
Breaking the Bottleneck in Cell Processing
The growth of cell therapy and diagnostics has outpaced the evolution of its core processing tools. Too often, the industry is forced to fit modern applications onto legacy platforms like centrifugation, which are harsh on cells, difficult to automate, and introduce unacceptable process variability. Overcoming this bottleneck demands a new approach built from the ground up for modern cell processing.
A Platform Built for Performance, Consistency, and Scale

Unmatched
Consistency & Quality
Our technology delivers consistent, high-quality output from variable biological inputs. By replacing manual steps with a closed, automated system, you can eliminate process variability and ensure superior cell yield, purity, and viability, run after run.

Engineered for
OEM Integration
Accelerate your time-to-market by leveraging our compact, continuous-flow modules as a validated subsystem. We provide a customizable engine, freeing your team to focus on instrument design, software, and workflow innovation.

Scalable by
Design
Precisely tune throughput from milliliters to liters per hour by multiplexing channels and modules to meet any need. Our modular approach provides a clear path from low-volume diagnostics to large-scale commercial manufacturing.
Designed for Integration
Manufactured for Scale
Integration-Ready Modules

Our compact sorter and concentrator modules are the foundational building blocks for your system. Featuring standard Luer-lock ports and a stackable design, they are engineered for simple, robust integration into any automated fluidic pathway.
Commercial-Scale Manufacturing

Through our partnership with Stratec, our ISO 13485-certified manufacturing process ensures quality and minimizes lot-to-lot variability. With a proven, high-volume supply chain in place, we de-risk your system launch and future growth.
Our Leadership & Advisors
BendBio is guided by a veteran executive team and world-class advisors. Their leadership provides deep, end-to-end expertise in two critical domains: the complete lifecycle of microfluidic technology from initial design and development to commercial-scale manufacturing – and the complex landscape of cell therapy, including research, process development, and successful commercialization. This unique fusion of engineering and biology ensures our solutions are not only innovative but also practical, scalable, and perfectly aligned with the real-world needs of our partners.
Executive Team
Advisors
Technical Deep Dive

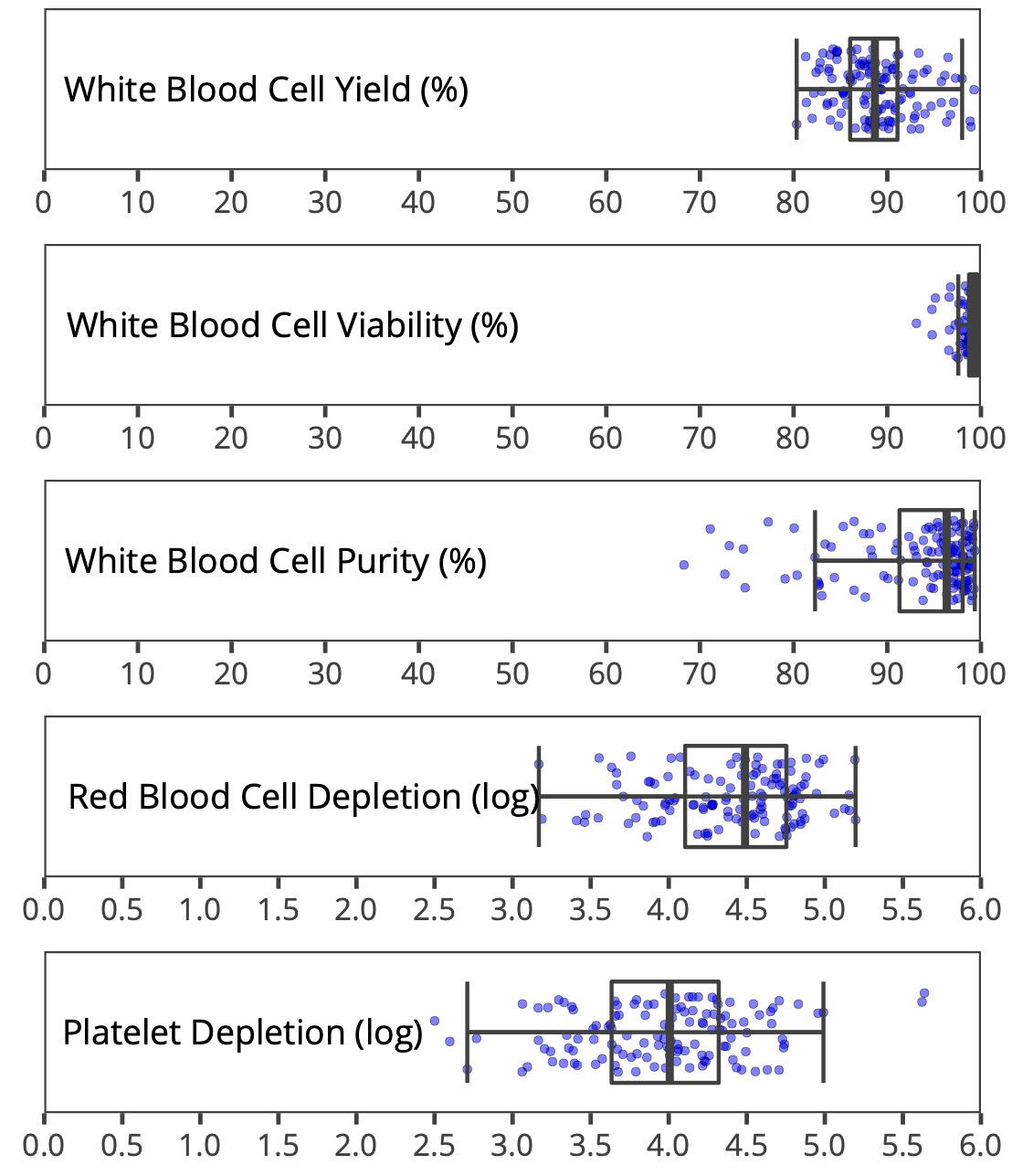
Data Highlight: Whole Blood Separation Performance
Validated Technology. Proven Results.
BendBio has engineered an inertial microfluidics platform, originally developed at Massachusetts General Hospital (MGH), to solve the key challenges of commercial-scale cell processing. The system delivers consistently high recovery, purity, and throughput. Its label-free mechanism is exceptionally gentle – with each cell spending just a fraction of a second undergoing processing – and requires no specialized buffers or reagents, preserving a pristine cell state. Critically, the platform transforms biologically variable input into highly consistent output, enabling the standardization of downstream processes. Our white paper validates this performance, achieving 90% yield, >99.99% removal of RBCs and platelets, and >99% cell viability in a demanding whole blood separation.
Get instant access to our white paper to see how this performance can break the bottlenecks in your own process.
Let's Build Your Next-Generation System
We partner with instrument manufacturers and life science companies to deliver superior, automated solutions. Contact us to discuss how the BendBio engine can power your platform.

Ravi Kapur, PhD
President, Founder
An executive, inventor, and innovator with a proven track record of driving innovation and commercialization in the biotechnology and life science industry. Ravi has built high-performance teams and has led change in competitive business environments. Over the past 25 years he has co-founded and led multiple venture-backed companies at the nexus of microfluidics and clinical medicine (Cellomics Inc. exited to ThermoFisher; Verinata Inc. exited to Illumina; MicroMedicine, AutoIVF, TellBio, San Simeon). Ravi holds an M.S. and Ph.D. in bioengineering from Clemson University.

Kyle Smith, PhD
CTO, Founder
A scientist and engineer with deep experience in microfluidics, computational modeling, and medical device development. Kyle invented and optimized the ultra-high-throughput microfluidic technology at Massachusetts General Hospital. He previously led R&D at MicroMedicine, Inc. and earned an M.S. in electrical engineering and computer science from MIT and a Ph.D. in biomedical and electrical engineering from the Harvard-MIT Division of Health Sciences and Technology.

Tom Barber, PhD
Head of Product Development & Operations, Founder
A scientist and engineer with over 15 years of experience in developing and translating microfluidic technologies into commercial products. Most recently, Tom led operations at MicroMedicine Inc. from the company’s inception to the launch of its disruptive Sorterra™ blood separation platform (Class I/CE marked medical device). Tom holds a Ph.D. from UC Berkeley/San Francisco’s joint graduate group in bioengineering.

Mehmet Toner, PhD
Technology Advisor, Founder
Andrus Benedict professor of bioengineering at the Massachusetts General Hospital, Harvard Medical School, and Harvard-MIT Health Sciences & Technology. Mehmet is internationally recognized for his research at the interface of nanotechnology, microsystems engineering, microfluidics, and applications in clinical medicine. He is a member of the National Academy of Medicine and the National Academy of Engineering.

Marcela Maus, MD, PhD
Scientific Advisor
Director of cellular immunotherapy at Massachusetts General Hospital and associate professor at Harvard Medical School. Her laboratory focuses on creating next-generation CAR T-cells (chimeric antigen receptor T-cells). Dr. Maus trained with Carl June, M.D. at the University of Pennsylvania and is a pioneer in the field of cancer immunology with particular expertise in T-cell immunotherapies.

Erik Rutjens, PhD
Industrial Advisor
CTO of a newly formed CAR T-cell therapy company, before that he was vice president, cell therapy operations at Enara Bio. Erik holds a Ph.D. in medicine (immunology) from the University of Leiden. He was previously at Novartis, where he played a critical role in the development of KYMRIAH, the first engineered T-cell therapy to be approved by the FDA.

Dominic Clarke, PhD
Industrial Advisor
Chief technical officer, cell & gene therapies at Discovery Life Sciences. Dominic holds a Ph.D. in cell & molecular biology. He is a recognized leader in the industry and was previously at HemaCare and Charles River Laboratories where he played a critical role in their global cell therapy business strategy.
Thank You for Your Inquiry
We will respond within 1–2 business days.
Download our white paper to learn more about how BendBio is addressing the challenges of cell therapy manufacturing and enabling next-generation cell processing systems.
Stay Connected
The best way to keep up with our latest data, technical insights, and company news is by following us on LinkedIn. Join our network of industry professionals building the future of automated cell processing.
Continue Exploring
Return to our home page to learn more about our mission, our technology, and the leadership team behind BendBio.
Get the White Paper
The Inertial Microfluidics Engine Enabling Next-Generation Cell Processing Platforms

Get the technical review instantly. Look under the hood at the core technology, performance data, and integration framework for your platform.
Thank You!
Your White Paper Is Ready for Download
Download to learn more about how BendBio is enabling next-generation cell processing systems.
Ready to Discuss Integration into Your System?
The white paper details the core technology. The next step is to discuss how our modules can be configured for your specific application and system requirements.
Stay Connected
Follow us on LinkedIn for the latest research, company updates, and insights into next-generation cell processing.
Continue Exploring
Return to our home page to learn more about our mission, our technology, and the leadership team behind BendBio.

The Inertial Microfluidics Engine for Your Next-Generation Platform
Download the white paper to look under the hood of our purpose-built engine for cell processing. Understand the core technology, its seamless integration capabilities, and the performance advantages it will bring to your platform.
Inside, you will find:
A deep dive into our core inertial microfluidics technology, explaining how the gentle, reagent-free process sorts and concentrates cells with remarkable efficiency, eliminating the harshness of legacy methods.
A framework to accelerate your time-to-market, covering our modular technology that scales from milliliters to liters per hour and a commercial-ready manufacturing process that de-risks your supply chain.
The performance advantages for your platform: eliminate process variability and deliver unmatched consistency, enabling you to provide superior cell yield, purity, and viability to your customers, run after run.
Get Instant Access

The Inertial Microfluidics Engine for Your Next-Generation Platform
Thank you for your interest at the trade show! Download the white paper to look under the hood of our purpose-built engine for cell processing. Understand the core technology, its seamless integration capabilities, and the performance advantages it will bring to your platform.
Inside, you will find:
A deep dive into our core inertial microfluidics technology, explaining how the gentle, reagent-free process sorts and concentrates cells with remarkable efficiency, eliminating the harshness of legacy methods.
A framework to accelerate your time-to-market, covering our modular technology that scales from milliliters to liters per hour and a commercial-ready manufacturing process that de-risks your supply chain.
The performance advantages for your platform: eliminate process variability and deliver unmatched consistency, enabling you to provide superior cell yield, purity, and viability to your customers, run after run.